29th September 2022
Cardiff Metropolitan University leads the way to develop a new and innovative Life saving Device to Monitor and Treat Heart Failure Patients
The Failing Heart and the Implantable Device.
Globally, approximately 26 million people are currently living with heart failure. This condition can deteriorate to a point where a patient’s heart can no longer provide the necessary function required to maintain life. It is at this point, where the very lucky will receive a suitable heart transplant. Unfortunately, due to shortages and suitability of donor hearts, the majority of patients will never receive a new heart. For these patients, mechanical circulator support systems such as Left Ventricular Assist Devices (LVADs) are often the last option to maintain life. Thankfully, these lifesaving implantable devices support the heart in pumping blood around the body and have significantly improved survival rates. However, many patients on LVAD support also present with increased rates of stroke and gastrointestinal (GI) bleeding.
What are Left Ventricular Assist Devices (LVADs)?
LVADs are continuous flow, non-pulsatile pumps. They are responsible for delivering blood flow into the patient’s end organs, such as the brain, gut and kidneys. However, the absence of a normal physiological pulse – a pulse you cannot palpate – has been identified as one of the main reasons for the elevated levels of debilitating stroke and GI bleeding associated with LVADs.
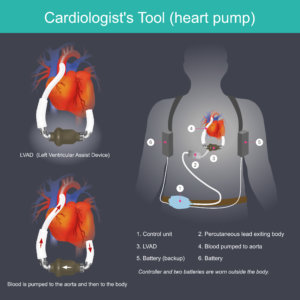
This electronic device is used to increase blood pressure from the heart to the aorta.
What is being done to help these patients?
For the past 10 years, cardiovascular researchers from Cardiff Metropolitan University, together with collaborating cardiology partners at the world-leading Columbia University Medical Center, New York Presbyterian Hospital, have made it their business to work together to better understand the blood flow and pressure profiles associated with risk to LVAD patients. The team are trying to understand what it is about these patients implanted with these specific pumps, their settings and their outputs that on the one hand improve survival rates, yet on the other, increase the risk of stroke and GI bleeding. This team of scientists, clinicians and an industrial partner from Germany have already secured approximately €250,000 from Horizon 2020, Marie Sklodowska Curie Action, to fund the successful “Haemodynamics Informing Treatment in patients implanted with Left Ventricular Assist Devices” (HIT-LVAD) trial. Their work in the area of mechanical circulatory support has been used and cited in the recent American Heart Association’s statement paper on “Device Therapy and Arrhythmia in Left Ventricular Assist Device therapy”, highlighting the clinical impact of the work. Such has been the ongoing interest in this research, that it secured a further $3.5 million in grant funding from the National Institute for Health (NIH) in 2022 to investigate “The neurovascular consequences of low pulsatility in patient’s on LVADs”, over the next five years.
Innovative technology to help measure and monitor patient’s blood pressure.
Furthermore, their ground-breaking research has resulted in the development of a novel device to measure blood pressure (BP) in patients implanted with the most recent LVADs, known as “the Heart Mate 3”, where measuring clinic/office BP is currently not possible. Partnerships between Cardiff Metropolitan University, Columbia University, New York, Leibniz University, Hanover, and an industrial partner in Germany are working closely to develop the world’s first device that can successfully measure BP in this patient cohort.
Professor Barry McDonnell, who leads the research team at Cardiff Metropolitan University said: “We are excited about the next stage of this research, particularly as it has the potential to globally impact the lives of so many LVAD patients in the future. The next objective of our team is to validate the accuracy of this new BP device, with the goal of helping clinicians to measure, monitor and treat high blood pressure and reduce stroke and GI bleeding risk in LVAD patients.”
If you would like to know more about this research please contact Professor Barry McDonnell at bmcdonnell@cardiffmet.ac.uk